por
John R. Fischer, Senior Reporter | January 18, 2019
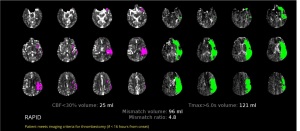
The RAPID platform can now be used
to select stroke patients who require
a blood clot procedure
The FDA has given its approval to iSchemaView’s RAPID neuroimaging platform to be used to identify stroke patients that require endovascular thrombectomy.
The platform is the only imaging solution to receive such clearance, which allows clinicians to apply CT- and MR-Perfusion to assess and determine if patients with known occlusion of the internal carotid artery or proximal middle cerebral artery should undergo a clot removal procedure.
"The addition of thrombectomy clearance makes RAPID even easier to use, especially by facilities and physicians with less expertise interpreting advanced imaging," Anil Singhal, M.D., SVP of worldwide operations at iSchemaView, told HCB News. "The flag, 'Patient meets imaging criteria for Thrombectomy' along with the clinical maps, simplifies the triage of these patients to the right center for the right treatment. RAPID results are ready in less than 2 minutes on the physician's phone via email and mobile app, as well as sent to the hospital's PACS."



Ad Statistics
Times Displayed: 74495
Times Visited: 5313 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
The RAPID platform is composed of an artificial intelligence framework made up of expert feature extraction and proprietary algorithms to support its offerings, which include CT perfusion; MR diffusion and perfusion; CT angiography; and CT ASPECT scoring. The solution’s modules offer custom notifications, custom thresholding and hypoperfusion, as well as cerebral blood volume indexes and golden hour thresholds.
Validated in more than 10 trials, RAPID has been deployed in landmark stroke studies that include SWIFT PRIME, EXTEND IA, EXTEND, DAWN and DEFUSE 3. The latter two successfully treated patients for up to 24 hours and led the American Heart Association and American Stroke Association to expand in their 2018 stroke guidelines the condition’s treatment window from six hours to 24 for eligible patients.
Its use is especially helpful to small providers, enabling clinicians to quickly and easily determine which patients to treat locally and which to send to thrombectomy centers urgently. Singhal says there are plans to diversify the providers that use it.
"RAPID works on all CT and MR scanners and is now available for scanners on Mobile Stroke Units (MSU)," said Singhal. "We're currently working on initiatives to have RAPID available in other scanning environments as well."
RAPID is installed on more than 4,000 scanners worldwide at over 1000 hospitals. More than 700 stroke centers
adopted the solution in 2018 alone, representing a year-to-date growth rate of 100 percent.
Back to HCB News

