por
John R. Fischer, Senior Reporter | February 07, 2023
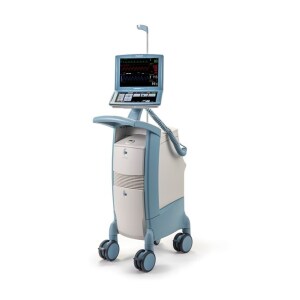
Getinge's Cardiosave Hybrid intra-aortic balloon pump (Photo courtesy of Getinge)
The FDA has weighed in on the recent recall of Getinge’s Cardiosave Hybrid and Rescue intra-aortic balloon pumps (IABP), labeling it as a Class I event, the most serious type, following complaints of four serious injuries and one death.
Getinge subsidiary Datascope recalled 4,454 Cardiosave solutions, designed to help the heart pump more blood, in December after receiving 134 complaints about it, including 12 unexpected shutdowns,
according to Reuters.
The cause of these shutdowns is a burst, leak or tear in the balloon that allows blood to enter the intra-aortic balloon pump during therapy, along with helium into the blood, causing the patient to experience blood loss.
As a result, patients are at risk for organ damage and death, particularly if they are critically ill and typically treated with Cardiosave. The release of helium in the blood can also damage organs, including the brain.
"Getinge is in the process of developing further instructions for use and may develop longer-term design improvements when it comes to ease of use and safety," wrote the company in an emailed statement.
The cost of the recall is not material, it says, but mitigating risks requires users to pay close attention to alarms for early identification of faulty balloons to stop blood from entering the device. Users should also periodically check the catheter tubing during therapy and stop pumping if they see blood,
according to Healthcare Dive.
Getinge also says users should immediately examine their inventories and remove Cardiosave Hybrid and Rescue IABPs with the defect from patient use.
The FDA previously categorized two Cardiosave recalls as Class I events in the second half of 2021, including one issue related to fluid entering the device and causing the pump to unexpectedly stop working. The issue was linked to one patient death, reported Healthcare Dive.
Prior to the current recall, the agency added Cardiosave devices to its list of product shortages, brought on by raw material shortages.
