por
John R. Fischer, Senior Reporter | October 10, 2019
GE Healthcare and Theragnostics partner on
development and commercialization of GalliProst
GE Healthcare and Theragnostics have struck up a global commercial partnership around the development and sale of the prostate-specific membrane antigen (PSMA) PET/CT imaging agent, GalliProst.
The scan indicates whether the disease is within the prostate gland, spread to the local lymph nodes or spread with more distant metastasis. Theragnostics will head development of the radiopharmaceutical, while GE will oversee all pre-approval commercial preparations, as well as all subsequent commercial and distribution tasks following the tracer’s approval.
"We believe GE is one of a few companies that provide a global reach into every market in the world. They also have the ability to access both generator-produced Ga-68 as well as cyclotron produced Ga-68," Greg Mullen, CEO of Theragnostics, told HCB News. "Ga-68 is the radioisotope in GalliProst that allows it to be visualized on the PET scanner."
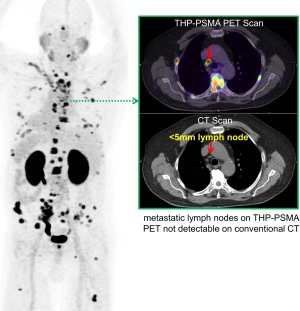
Over 1.2 million new cases of prostate cancer were diagnosed at more than 350,000 facilities worldwide in 2018, making it the fourth most widespread type of cancer. A blood test or biopsy is used to detect prostate cancer, after which patients are usually referred for a PET/CT scan so that radiologists can examine the tumor, lymph nodes and any metastasis to determine the best course of treatment.
Derived from Gallium-68, the new tracer provides clinicians with "heatmap-style" images that show the precise location and intensity of PSMA, which is expressed on the surface of prostate cancer cells. A phase II clinical study that met its primary and secondary endpoints showed that clinicians who used GalliProst ended up modifying treatment plans for one third of newly diagnosed prostate cancer patients — and over 50 percent with biochemically recurrent disease. This change in patient management rose to 75 percent in a post-radical radiotherapy environment.
"The scan provides the physician with an indication as to whether the disease is within the prostate gland, spread to the local lymph nodes or has spread with more distant metastasis," said Mullen. "Conventional imaging (CT or bone scan) has difficulty in detecting small volume disease outside of the prostate. Therefore, GalliProst can help determine whether curative therapy (surgery or targeted radiotherapy) or systemic treatment (hormone/chemotherapy) is the most appropriate treatment."
The agreement follows another orchestrated last month in which AstraZeneca
granted Theragnostics a license to operate globally in the diagnostic field of certain selected radionuclide-labelled PARPi (Poly (ADP-Ribose) Polymerase inhibitors), with an option to an exclusive license for freedom to operate globally in the therapeutic field of certain selected radionuclide-labelled PARPi.
Both GE Healthcare and Theragnostics will be on hand to discuss their findings from October 12-16 at the European Association of Nuclear Medicine 2019 Congress (EANM) in Barcelona.
