por
Lauren Dubinsky, Senior Reporter | June 23, 2015
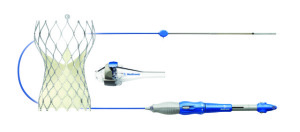
Medtronic's CoreValve Evolut R System
Transcatheter aortic valve replacement (TAVR) has become a go-to treatment option for high-risk patients with severe aortic stenosis who are unable to undergo surgery, but physicians are now searching for technology advancements to deliver even better patient outcomes.
Medtronic announced today that its CoreValve Evolut R System received FDA approval to be used for TAVR in severe aortic stenosis patients who are at high or extreme risk for surgery. It is the first and only recapturable and repositionable device available in the U.S., according to the company.
"Clinical data have shown the best patient outcomes are achieved when the valve is properly positioned,” Dr. Mathew Williams, chief of adult cardiac surgery and director of the Structural Heart and Valve Program, and Director of Interventional Cardiology at the NYU Langone Medical Center. “The advancement of recapturability with Evolut R gives physicians more confidence during the procedure and provides advantages that are non-existent in other TAVR systems."



Ad Statistics
Times Displayed: 6679
Times Visited: 20 Brand-New FDA-cleared Advanced Ultrasound Medical Device available for sale or lease to Wound Care Centers or any other Medical Facilities.The Arobella 1000D is designed for non-contact or debridement ultrasound wound healing therapy, or any other wounds
Aortic stenosis is a condition where the aortic valve narrows and limits blood flow from the aorta to the rest of the body. If left untreated, the patient may experience serious heart problems including heart failure and death.
In March, Medtronic’s other heart valve, CoreValve System, became the first TAVR system to receive approval in the U.S. for valve-in-valve procedures in patients whose surgical aortic heart valve failed. To date it has been implanted in over 75,000 patients in 60 countries.
At the American College of Cardiology conference this year, two-year data from the High Risk Study of the CoreValve U.S. Pivotal Trial was presented, which showed that more patients who underwent TAVR with the CoreValve System survived after two years than patients who underwent surgical aortic valve replacement.
The new system is composed of the CoreValve Evolut R transcatheter valve and the EnVeo R Delivery System, which features an InLine Sheath that reduces the profile to the lowest on the market. A smaller profile means that patients with smaller vessels can also be treated now.
The 23 mm, 26 mm and 29 mm sizes of the CoreValve Evolut R transcatheter valve and the CoreValve EnVeo R Delivery Catheter System are available for use in the U.S. It is also available in Europe and other countries that recognize the CE mark.
