por
Brendon Nafziger, DOTmed News Associate Editor | June 28, 2011
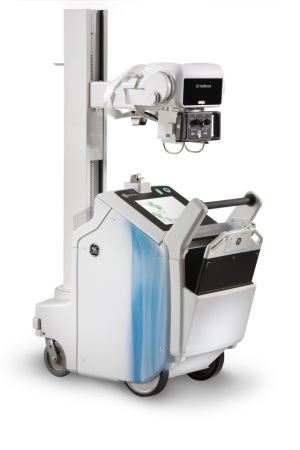
Optima XR220amx
(Credit: GE)
GE Healthcare said last week it launched a trio of new mobile X-ray systems. The Optima XR220amx, Optima XR200amx and Brivo XR285amx have all received Food and Drug Administration 510(k) clearance, the Waukesha, Wis.-based company said.
The battery-powered, general-purpose new platforms are more compact than earlier versions, GE said, and are designed for point-of-care scans of patients unable to get to the radiographic room. All three also feature an integrated, 15-inch touchscreen user interface.
The digital Optima XR220amx works with the company's FlashPad digital wireless detector, which received FDA clearance last November.



Ad Statistics
Times Displayed: 14289
Times Visited: 25 Final days to save an extra 10% on Imaging, Ultrasound, and Biomed parts web prices.* Unlimited use now through September 30 with code AANIV10 (*certain restrictions apply)
In a spec sheet, GE dished out more details on Optima XR220amx's detector. Its battery can take up to 45 exposures per hour, and has enough power for three hours of use on a single charge, GE said. The 10-pound device can also support up to 330 pounds of distributed load for bariatric applications.
Another of the three models, the Optima XR200amx, is what GE calls "digital-ready," meaning it's designed to work without a digital detector, but the internal image-processing hardware is set up so it’s easily upgraded to digital, GE said.

